Exercise : Solution of Questions on page Number : 66
Answer 1:
(a) Methyl orange: Methyl orange changes its colour to yellow when added to a base or an alkali.
(b) Litmus paper: Red litmus paper turns blue, whereas blue litmus paper remains blue in a base or an alkali.
(c) Red cabbage extract: Red cabbage extract turns greenish-yellow in basic solutions.
Answer 2:
(a) Litmus paper: Blue litmus paper turns red to an acid, whereas red litmus paper remains red.
(b) Methyl orange: Methyl orange changes to red when added to an acid.
Answer 3:
Red litmus paper is red in an acid solution and turns blue in a basic solution.
Answer 4:
Phenolphthalein is pink in an alkaline solution but turns colourless in an acidic solution.
Answer 5:
Basic solutions subdue the pungent smell of onion, however acidic solutions have no such effect. Hence we can say that, the cloth strip must have been treated with a basic solution.
Answer 6:
When a basic solution is added to vanilla extract, it destroys the characteristic smell of vanilla. Conversely, it is retained in an acidic solution. Hence, the given solution is a base.
Answer 7:
Hydrogen gas is liberated when an active metal reacts with hydrochloric acid.
For testing the presence of hydrogen gas, bring a burning candle near the mouth of the test tube in which the reaction is taking place. If the gas burns with a popping sound, then it can be concluded that it is hydrogen.
Answer 8:
Carbon dioxide gas is liberated when dilute hydrochloric acid reacts with sodium hydrogen carbonate.
When the gas liberated in the reaction is passed through lime water, lime water turns milky or a white precipitate of calcium carbonate is formed. This confirms that the gas is carbon dioxide.
Answer 9:
The names and formulae of two strong acids and two weak acids are as follows.
(a) Strong acids:
1. Hydrochloric acid: HCl
2. Sulphuric acid: H2SO4
(b) Weak acids:
1. Formic acid: HCOOH
2. Acetic acid: CH3COOH
Answer 10:
(a) Citric acid: Lemons and oranges
(b) Oxalic acid: Tomatoes
(c) Lactic acid: Sour milk
(d) Tartaric acid: Unripe grapes and tamarind
Answer 11:
Animal sting containing formic acid: Ant sting
Plant sting containing formic acid: Nettle leaf sting.
Answer 12:
When the solution of an acid is diluted, the concentration of hydronium ions decreases because the total volume of the solution increases on addition of water.
Concentration of hydronium ions=Volume of solute (acid)Volume of solution.
Answer 13:
(a) Sulphuric acid + Zinc → Zinc Sulphate solution + Hydrogen gas
dil. H2SO4(aq) + Zn(s) → ZnSO4(aq) + H2(g)
(b) Hydrochloric acid + Magnesium → Magnesium chloride solution + Hydrogen gas
dil. 2HCl(aq) + Mg(s) → MgCl2(aq) + H2(g)
(c) Sulphuric acid + Aluminium → Aluminium sulphate solution + Hydrogen gas
dil. 3H2SO4(aq) + 2Al(s) → Al2(SO4)3(aq) + 3H2(g)(d) Hydrochloric acid + Iron → Iron(III) chloride solution + Hydrogen gas
dil. 6HCl(aq) + 2Fe(s) → 2FeCl3(aq) + 3H2(g).
Exercise : Solution of Questions on page Number :67
Answer 14:
(a) Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
(b) Na2CO3(s) + 2HCl(aq) → 2NaCl(aq) + CO2(g) + H2O(l)
(c) NaHCO3(s) + HCl(aq) → NaCl(aq) + H2O(l) + CO2(g)
(d) NaOH(aq) + HCl(aq)→ NaCl(aq) + H2O(l)
(e) CuO(s) + 2HCl(aq) → CuCl2(aq) + H2O(l)
Answer 15:
(a) Acids have a sour taste and they turn blue litmus to red.
(b) Substances do not show their acidic properties without water.
(c) Acids produce hydronium ions on dissolving in water.
(d) Those substances whose smell (or odour) changes in acidic or basic solution are called olfactory indicators.
(e) Onion and vanilla extract are olfactory indicators.
Answer 16:
(a) Indicators are substances that show a distinct change (colour or odour) simultaneous to a change in concentration of ions such as H+ and OH– ions.
Examples: Methyl orange, phenolphthalein and litmus solution.
(b) A purple dye is extracted from a plant called lichen. This dye is called a litmus solution and is commonly used as an indicator.
(c) When turmeric paper is put into an alkaline solution, it turns red.
Answer 17:
Olfactory indicators are those indicators that change their odour when added to an acidic or a basic solution. Onion and vanilla are examples of olfactory indicators.
The characteristic smell of an olfactory indicator is lost when it is added to a sodium hydroxide solution.
Answer 18:
(a) When a metal is treated with an acid, it liberates hydrogen gas from the acid and combines with the remaining part of the acid to form a compound called a salt.
Chemical equation:
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
(b) Hydrogen gas is usually liberated when an acid reacts with an active metal.
For testing the presence of hydrogen gas, bring a burning candle near the test tube in which reaction has taken place. If the gas burns with a popping sound, the gas is hydrogen.
Answer 19:
While diluting an acid, the acid should always be added slowly to water with constant stirring and in small amounts. The dilution of an acid is an exothermic reaction. So, when we add acid to water, the evolved heat gets absorbed by the water that is in bulk. But when we add water to acid, a large amount of heat is evolved, the water turns into vapour and the acid splashes onto our clothes or face, causing severe burns.
Answer 20:
When an acid is added to a metal hydrogen carbonate, carbon dioxide gas is liberated and metal salt and water are formed.
This reaction can be represented as follows.
Acid + Metal hydrogen carbonate → Salt + Water + Carbon dioxide gas
Example: When sodium hydrogen carbonate is treated with hydrochloric acid, carbon dioxide gas is liberated and sodium chloride is formed.
The reaction that takes place can be written as follows:
NaHCO3(s) + HCl(aq) → NaCl(aq) + H2O(l) + CO2(g)
Answer 21:
(a) When sodium carbonate is treated with dilute hydrochloric acid, sodium chloride is formed and carbon dioxide gas is liberated.
Na2CO3(s) + 2HCl(aq) → 2NaCl(aq) + CO2(g) + H2O(l)
(b) Carbon dioxide gas is liberated when dilute hydrochloric acid reacts with sodium carbonate.
When the gas, liberated during the reaction, is passed through lime water, the lime water turns milky or a white precipitate of calcium carbonate is formed. This confirms that the gas is none other than carbon dioxide.
Answer 22:
When an acid reacts with a base, it nullifies the effect of the base. Such a reaction between an acid and a base to give a salt and water is known as a neutralisation reaction.
The reaction can be represented as given below.
Acid + Base → Salt + Water
For example, when sodium hydroxide is treated with hydrochloric acid, the following reaction takes place,
NaOH(aq) + HCl(aq) → H2O(aq) + NaCl(aq)
This kind of reaction is called a ‘neutralisation reaction’.
Answer 23:
When an acid is treated with a metal oxide, a salt and water are obtained.
The general reaction can be written as follows:
Acid + Metal oxide → Salt + Water
For example, when a small amount of copper oxide is added to sulphuric acid, colour of the solution becomes blue and the copper oxide dissolves. The blue colour of the solution indicates the formation of copper (II) sulphate.
The reaction taking place can be written as follows:
CuO(s) + H2SO4(aq) → CuSO4(aq) + H2O(l).
Answer 24:
(a) Organic acids: An organic acid is an acid that is found naturally in plants and animals. Generally, organic acids are weak acids and do not dissociate completely in water.
Mineral acids: Mineral acids are man-made and are derived from one or more inorganic compounds. They are also known as inorganic acids. They range from acids of great strength to those that are very weak.
(b) Here are a few common examples.
Organic acids
Lactic acid
Citric acid
Mineral acids
Hydrochloric acid (HCl)
Nitric acid (HNO3)
(c) Uses of mineral acids in industry:
Mineral acids are used in many sectors of the chemical industry for the synthesis of various chemicals.
Dilute solutions of hydrochloric acid are used for removing the deposits from inside boilers, with precautions to prevent the corrosion of the boilers by the acid.
They are also used in the processing of leather, purification of common salt, construction of buildings, etc.
Answer 25:
Strong acids are those that completely dissociate into their ions when mixed with water, whereas weak acids are those that dissociate only partially into their ions when added to water.
Strong acids – HCl, H2SO4, HNO3
Weak acids – CH3COOH, H2CO3, H2SO3
Answer 26:
HCl, H2SO4, HNO3, etc., dissociate in aqueous solutions to give H+(aq) ions, which are a vital part of an acid and determine the acidic property of acids.
C6H12O6 (glucose) and C2H5OH (alcohol) do not dissociate in aqueous solutions even though they contain hydrogen atoms.
Hence, HCl, H2SO4, HNO3, etc., show acidic character in aqueous solutions, whereas solutions of compounds such as C6H12O6 (glucose) and C2H5OH (alcohol) do not show acidic character.
Answer 27:
When an acid reacts with a base, the acid nullifies the effect of the base, and conversely, the effect of an acid is nullified by a base. Such a type of reaction between an acid and a base to give a salt and water is known as a neutralisation reaction.
The reaction can be represented as follows:
Acid + Base → Salt + Water
For example, when sodium hydroxide is treated with hydrochloric acid, sodium hydroxide and hydrochloric acid neutralise each other to form common salt (NaCl) and water. The reaction can be given as below:
NaOH (aq) + HCl (aq) → NaCl (aq) + H2O (l)
Answer 28:
Curd and other foodstuffs (such as lemon juice) are acidic in nature. Acids react with metals to give hydrogen gas. Hence, when these acidic foodstuffs are kept in metal containers, hydrogen gas is liberated, and it spoils the food and makes it inedible.
Answer 29:
(a) When an acid is added to a base, the acid nullifies the effect of the base, and conversely, the effect of an acid is nullified by a base to give a salt and water. Such a type of reaction between an acid and a base to give a salt and water is known as a neutralisation reaction.
The reaction can be represented as follows.
Acid + Base → Salt + Water
For example, when sodium hydroxide is treated with hydrochloric acid, the reaction can be written as follows.
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)
(b) Dry HCl gas does not undergo dissociation to form ions, because of the absence of an aqueous medium, whereas the colour of litmus paper changes only in the presence of ions. Hence, dry HCl gas does not change the colour of dry litmus paper.
(c) A phenolphthalein indicator turns pink when added to an alkali (such as sodium hydroxide).
Answer 30:
(a) The acidic behaviour of a substance is due to the presence of H+ ions. As acids dissociate into their ions only in the presence of water, the acidic character of acids cannot be seen in the absence of water.
(b) Acids dissociate to release H+ ions and an anion, when dissolved in water. When electric current is passed through an aqueous solution, the ions start moving toward oppositely charged terminals of the battery, henceforth, conducting electricity.
(c) Distilled water is the purest form of water. The process of distillation makes the water free from all the ionic species, whereas rainwater consists of plenty of impurities. Thus, it contains numerous ions such as H+, CO3-, SO42- etc. Therefore, rainwater conducts electricity.
Exercise : Solution of Questions on page Number : 68
Answer 31:
(a) Metal carbonates react with acids to give a corresponding salt, carbon dioxide and water.
The reaction can be represented as follows.
Metal carbonate + Acid → Salt + Carbon dioxide + Water.
For example, when sodium carbonate reacts with dilute hydrochloric acid, sodium chloride, water and carbon dioxide gas are produced. The reaction occurs as follows.
Na2CO3(s) + 2HCl(aq) → 2NaCl(aq) + H2O(l) + CO2(g)
(b) When carbon dioxide gas is passed through lime water, the lime water turns milky. This confirms that the gas liberated in the reaction is none other than carbon dioxide.
(i) The reaction that occurs when carbon dioxide is passed through lime water for a short time can be represented as follows:
Ca(OH)2(aq) + CO2(g) → CaCO3(s) + H2O(l)
When carbon dioxide gas is passed through lime water, it turns the lime water milky. This happens because of the formation of white precipitate of calcium carbonate.
(ii) When carbon dioxide is passed through lime water for a considerable time (in excess), the following reaction occurs with the formation of calcium bicarbonate:
CaCO3(s) + H2O(l) + CO2(g) → Ca(HCO3)2(aq)
When carbon dioxide gas is passed through lime water for a considerable time, a white precipitate of calcium carbonate is dissolved. This happens because of the formation of calcium hydrogen carbonate, which is soluble in water.
Answer 32:
Procedure:
Take about 1 g of sodium chloride (NaCl) in a clean and dry test tube.
Add some concentrated sulphuric acid (H2SO4).
A gas is produced and comes out through the delivery tube.
Test the gas evolved with dry and wet strips of litmus paper.
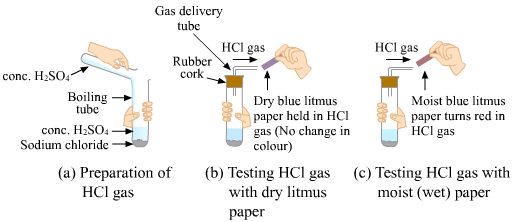
Observation:
(i) When the gas evolved is passed through dry litmus paper, there is no change in the colour of litmus paper.
(ii) However, when the gas is passed through wet blue litmus paper, the paper turns red, which indicates that the gas evolved is an acid, HCl.
Conclusion:
This activity concludes that hydrogen ions in HCl are produced in the presence of water because only wet blue litmus turns red.
Answer 33:
(a) All acids have hydrogen as a common element in them.
Examples: Hydrochloric acid (HCl), sulphuric acid (H2SO4), nitric acid (HNO3).
(b) Activity to show that alcohol and glucose also contain hydrogen but are not acids:
Procedure:
i) Take samples of alcohol, glucose and hydrochloric acid.
ii) Take a 100 ml beaker. Take a cork and fix two nails on it.
iii) Place the cork inside the beaker as shown in the figure.
iv) Connect the nails to two terminals of a 6-volt battery through a bulb and a switch.
v) Now pour some dilute hydrochloric acid in the beaker so that the nails and the cork are immersed in it.
vi) Switch on the current flow.
vii) Repeat the same with alcohol and glucose.
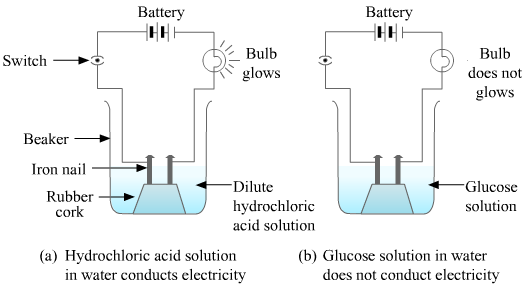
Observation :
The bulb starts glowing in the case of acid, whereas it does not glow in the case of glucose and alcohol solutions. Glowing of the bulb signifies that there is a flow of electric current through the solution. The electric current is carried through the solution by ions.
Conclusion:
Acids dissociate in aqueous solutions to give H+(aq) ions, which determine their acidic property. Glucose and alcohol do not dissociate and do not furnish H+ ions in aqueous solutions even though they contain hydrogen atoms. Hence, HCl shows acidic character in aqueous solutions, whereas solutions of compounds such as C6H12O6 (glucose) and C2H5OH (alcohol) do not show acidic character.
Answer 34:
(d) 16 mL
It is given that 10 mL of a solution of NaOH is completely neutralised by 8 mL of a solution of HCl. Hence, 20 mL of the solution of NaOH, which is double the amount taken earlier, will require double the solution of HCl taken earlier, i.e., 16 mL, to be completely neutralised.
Answer 35:
(c) antacid
Indigestion is caused by the formation of excess acid in the stomach. An antacid is a medicine that is generally a base or a basic salt that neutralises acidity in the stomach.
Answer 36:
(c) H2SO4
Marble chips contain calcium carbonate (CaCO3), which reacts with sulphuric acid (H2SO4) to form carbon dioxide gas (CO2). Carbon dioxide gas, when passed through lime water [Ca(OH)2], turns it milky.
Answer 37:
(d) carbonic acid
Carbonic acid H2CO3 is formed when CO2 is dissolved in water. Hence, carbonic acid is an inorganic or mineral acid. Ethanoic acid, formic acid and citric acid are found naturally.
Answer 38:
(b) they feel soapy
Acids have a sour taste, they turn blue litmus paper red and their pH is less than seven. But they do not feel soapy.
Answer 39:
(c) litmus and methyl orange
Litmus and methyl orange turn red in an acid solution.
Answer 40:
(c) baking soda
Baking soda, being a base, neutralises the excess acid formed in the stomach and cures the discomfort caused by indigestion.
Answer 41:
(d) have sour taste
Vinegar and curd are acidic. They both have a sour taste.
Answer 42:
(c) phenolphthalein
Phenolphthalein produces a pink colour in an alkali solution.
Answer 43:
(d) HCl
Zinc granules react with HCl to give hydrogen gas, which burns with a ‘pop’ sound.
Answer 44:
(a) X is carbon dioxide gas (CO2).
Y is calcium carbonate (CaCO3).
Z is calcium hydrogen carbonate (calcium bicarbonate) [Ca(HCO3)2].
(b)
(i) When a piece of limestone or calcium carbonate is treated with dilute hydrochloric acid, carbon dioxide gas (CO2 , here given as X) is produced.
CaCO3(s) + 2HCl(aq) → 2NaCl(aq) + H2O(l) + CO2(g)
(ii) When carbon dioxide (X), liberated in the reaction is passed through lime water, the lime water turns milky or a white precipitate of calcium carbonate (CaCO3 , Here given as Y) is formed.
Ca(OH)2(aq) + CO2(g) → CaCO3(s) + H2O(l)
(iii) When excess of carbon dioxide is passed through lime water, a solution of calcium hydrogen carbonate [Z, Ca(HCO3)2] is formed.
CaCO3(s) + H2O(l) + CO2(g) → Ca(HCO3)2(aq)
Answer 45:
Baking soda can be suggested as a remedy to the person suffering from acidity. Acidity is caused by the production hydrochloric acid in the stomach, and excess of acid causes pain and burning. Baking soda (NaHCO3) is basic in nature and it neutralizes the HCl produced in the stomach, bringing relief.
NaHCO3 (s)+ HCl (aq) → NaCl (aq) + CO2 (g)+ H2O(l)
Exercise : Solution of Questions on page Number : 69
Answer 46:
(a) When cupric oxide is treated with dilute hydrochloric acid, copper(II)chloride (blue-green salt solution) and water are obtained.
(b) CuO(s) + 2HCl(aq) → CuCl2(aq) + H2O(l)
(c) The reaction between a metal oxide and hydrochloric acid is similar to the neutralisation reaction in which, after the reaction, a salt and water are formed. So, we can conclude that copper oxide is basic in nature.
Answer 47:
(a) The curry contains turmeric which works as a natural indicator.
(b) When the shirt with a yellow stain of curry is washed with soap, the stain turns reddish brown because soap is basic and turmeric gives a reddish-brown colour when reacts with a base. When rinsed with plenty of water, the reddish-brown stain turns yellow again due to removal soap.
(c) Soap is basic in nature because it turns the yellow turmeric stain reddish brown.
Answer 48:
We have been provided with three test tubes, containing distilled water, an acidic solution and a basic solution. The test tube containing distilled water will not bring about any change in the colour of the blue litmus paper. Now, when you dip the same litmus paper in the test tube containing the acidic solution, it will turn the blue colour of the litmus paper to red. The basic solution in the third test tube will have no effect on blue litmus paper but will revert back the colour of litmus paper to blue which was earlier turned red by the acidic solution.
Answer 49:
Sodium hydrogen carbonate (baking soda) is used as an antacid because it neutralises excess acid present in the stomach. It reacts with dilute hydrochloric acid to produce carbon dioxide gas, which is used in fire extinguishers.
Hence, substance X is sodium hydrogen carbonate (baking soda) and Y is carbon dioxide gas.The reaction between sodium hydrogen carbonate and hydrochloric acid gives sodium chloride, carbon dioxide and water.
HCl(aq) + NaHCO3(s) → NaCl(s) + H2O(l) + CO2(g)
Answer 50:
Metal carbonates react with acids to give a corresponding salt, carbon dioxide and water.
The reaction can be represented as follows:
Metal carbonate + Acid → Salt + Carbon dioxide + Water
Example:
Na2CO3(s) + 2HCl(aq) → 2NaCl(aq) + H2O(l) + CO2(g)
However, the neutralisation of an oxide or a hydroxide with an acid gives only a salt and water.
Metal oxide + Acid → Salt + Water
Example:
Na2O + 2HCl → 2NaCl + H2O
Metal hydroxide + Acid → Salt + Water
Example:
NaOH + HCl → NaCl + H2O
Answer 51:
(a) When a base is added to an acid, it nullifies the effect of the acid and removes the H+ ions from it and turns them into water. During a neutralisation reaction, the H+ ions of an acid are converted into water.
The reaction can be represented as follows.
Acid + Base → Salt + Water
For example, when sodium hydroxide is treated with hydrochloric acid, sodium chloride and water are produced.
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)
(b) The temperature of the solution increases, because a neutralisation reaction is an exothermic reaction in which heat is produced.
Exercise : Solution of Questions on page Number : 79
Answer 1:
(a) When zinc granules are treated with hydrochloric acid, zinc chloride is formed and hydrogen gas is liberated.
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
(b) Zinc reacts with sodium hydroxide, giving sodium zincate and hydrogen gas.
Zn(s) + 2NaOH(aq)→ Na2ZnO2(aq) + H2(g)
Answer 2:
Water-soluble bases are commonly known as alkalies. An alkali is a unique base that dissolves in water without any chemical reaction. All alkalies are bases, whereas only some bases are alkalies.
Answer 3:
An alkali is a unique base that is soluble in water and forms hydroxide ions (OH-) when dissolved in water.
Answer 4:
Tooth enamel is made up of calcium phosphate and is the hardest part of the body. When the pH in the mouth goes down and becomes 5.5, it is strong enough to corrode the enamel of the tooth. The mouth turns acidic because the bacteria present in it produce acids by breaking down sugar and food particles left behind in it after eating.
Answer 5:
A neutral solution has a pH value of 7. It is free of ions such as H+ and OH–.
Answer 6:
A solution with a pH of 2 is more acidic than a solution with a pH of 6, because an acidic solution has a pH value of less than 7. As the pH value decreases from 7, the acidity of a solution increases because there is an increase in the concentration of hydrogen ions (H+ ions).
Answer 7:
A solution with a pH of 11 is more basic than a solution with a pH of 8. This is because a pH value greater than 7 indicates a basic solution. As the pH value increases from 7 to 14, there is an increase in the concentration of OH-, which increases the strength of the solution.
Answer 8:
The scientist, Soren Sorenson developed the pH scale.
Answer 9:
A universal indicator shows different colours at different concentrations of hydrogen ions in a solution and tells us how strong or weak an acid or a base is.
Answer 10:
Soil B, with a pH of 4.5, is very acidic and is thus not suitable for the normal growth of plants. Therefore, to reduce its acidity, soil B should be treated with powdered chalk (CaCO3), which is a base.
Answer 11:
A universal indicator shows different colours at different concentrations of hydrogen ions in a solution and can be used for testing the pH of a solution.
Answer 12:
| SUBSTANCE | COLOUR CHANGE |
| Potassium hydroxide | Dark purple |
| Soda water | Orange yellow |
| Sulphuric acid | Red |
Answer 13:
HCl, with a pH of 1, imparts a red colour when treated with a universal indicator. As the pH value decreases from 7, the acidity increases, as there is an increase in the concentration of hydrogen ions (H+). So, the given acid is very strong.
Answer 15:
Solution A, being acidic, with a pH value of 3.0, will turn litmus solution from blue to red, and solution B, being basic, with a pH value of 9.5, will turn phenolphthalein from colourless to pink.
Answer 16:
Drink Q will have a pH value of 9, because basic solutions have a pH greater than 7 on a pH scale.
Answer 17:
As per the given data, solution X has a pH value of 4 (which is less than 7) and will give an acidic reaction. Solution Y has a pH value of 8 (which is greater than 7) and will give an alkaline reaction.
Answer 18:
(a) Acids have a pH less than 7.
(b) Alkalis have a pH greater than 7.
(c) Neutral substances have a pH of 7.
(d) The more acidic a solution, the lesser the pH.
(e) The more alkaline a solution, the greater the pH.
Answer 19:
Usually, fresh milk has a pH of 6. When fresh milk changes into curd, its pH value decreases because of an increase in the acidity of the milk. Bacteria usually change fresh milk to curd by producing lactic acid. Fresh milk that has changed into curd is sour, because its acidity has increased. Hence, the pH value of fresh milk becomes less than 6 when it changes into curd.
Answer 20:
(a) A universal indicator is a combination of different dyes that show colour change with solutions of different pH values on the complete pH scale.
A universal indicator paper is used to differentiate between acids and bases. It also differentiates between strong acids and bases and between weak acids and bases on the basis of the intensity of colour change.
(b) When one or two drops of an acid or a base solution are added to a universal indicator paper, it generates a new colour. By comparing the colour with the pH scale, the pH of the added solution is known. From the pH value, we can identify the strength of the acid or base solution.
(c) A neutral substance has a pH value of 7. When we add a few drops of a universal indicator to a test tube of water (which is a neutral substance), we get a green colour, indicating the neutrality of water.
Answer 21:
(a) Methanoic acid is injected into the skin of a person during an ant sting.
(b) Methanoic acid is injected into the skin during a nettle-leaf-hair sting.
Both an ant sting and a nettle-leaf-hair sting are acidic, as methanoic acid is injected into the body, which causes severe pain and an itching sensation. Applying a baking soda solution (a base) on the skin soothes the pain by neutralising the acid. Thus, the effect of these stings is neutralised.
Exercise : Solution of Questions on page Number : 80
Answer 22:
(a) When the pH of the mouth falls to around 5.5, tooth decay begins. As the acidity in the mouth increases, the acid reacts with the tooth enamel and causes tooth decay.
Tooth decay can be prevented by brushing with toothpastes (which are basic and neutralise the acid formed) regularly and by cleaning the mouth carefully after meals.
(b) Because of acid rain (when the pH of rainwater is 5.6), the acidity of river and lake water increases. Aquatic animals find it very difficult to survive in water with low pH levels, and they die.
To neutralise this excess acidity in lakes and rivers caused by acid rain, calcium carbonate, a base, is frequently mixed with the water.
Answer 23:
(a) When a bee stings a person, it injects an acidic solution into the person’s skin. This causes severe pain and irritation. The remedy is to apply a baking soda solution (sodium hydrogen carbonate solution, which is a base). The solution soothes the pain by neutralising the acid.
(b) When a wasp stings a person, it inserts an alkaline solution into the person’s skin. This causes severe pain and irritation. The remedy is to apply a weak acid, such as vinegar, to the wound. The acid neutralises the alkaline solution and soothes the pain.
Answer 24:
(a) When a bee stings a person, it injects an acidic solution into the person’s skin, which causes severe pain and irritation. This acid has to be neutralised by a base and not an acid. If we apply vinegar, which is a weak acid, the pain worsens. Hence, it is wrong to treat a bee sting with vinegar.(b) When a wasp stings a person, it inserts an alkaline solution into the person’s skin, which causes severe pain and irritation. The alkali has to be neutralised by an acid and not a base. If we apply a baking soda solution, which is a weak base, it worsens the pain. Hence, it is wrong to treat a wasp sting with a baking soda solution.
Answer 25:
(a) The pH of a solution signifies whether it is an acid or a base and also its strength. Solutions with a pH greater than 7 are considered basic or alkaline, solutions with a pH less than 7 are acidic, and solutions with a pH of 7 are considered neutral.
Solution B with the lower pH value of 4 is highly acidic.
(b) If the pH of the soil is 4.2, then it is highly acidic. Hence, the soil should be treated with chemicals (bases) such as quick lime (calcium oxide) or slaked lime (calcium hydroxide).
Answer 26:
(a) (i) Acidic solutions: A with pH = 0, D with pH = 3, C with pH = 6
(ii) Basic solutions: F with pH = 8, B with pH = 11, E with pH = 13
(b) The acids or alkalis used to make the following products are given against them.
(i) Car batteries – Sulphuric acid
(ii) Explosives – Nitric acid (in explosives such as TNT, or trinitrotoluene)
(iii) Soaps – Sodium hydroxide
(iv) Fertilisers – Nitric acid, sulphuric acids
Answer 27:
a) A cold drink with a pH value of 5 is acidic. Therefore, blue litmus turns red and red litmus remains as it is.
b) Acids have a pH value of less than 7. Lower the pH value, the stronger the acid. Hence, the acids in the increasing order of their strengths are as follows: A < C < B.
Answer 28:
Calcium oxide, calcium hydroxide and calcium carbonate are all bases (pH > 7), and hence, they must be used only in cases where the soil is too acidic (pH < 7). They help in neutralising the acidity of the soil and assist in bringing the pH closer to 7.
Answer 29:
Dilute hydrochloric acid is the acid produced in our stomach. Sometimes, excess acid is produced in the stomach for various reasons such as overeating. When this happens, the excess generation of acid in the stomach causes indigestion with pain and discomfort. This can be cured by the intake of antacids, which are weak bases. They neutralise the excess acid produced in the stomach, providing relief from indigestion and relieving the pain.
Answer 30:
The soil can be treated with bases such as quick lime (calcium oxide) or slaked lime (calcium hydroxide) to reduce its acidity.
These bases react with the excess acid present in the soil and neutralise its acidity. This increases the pH of the soil, which makes it suitable for the growth of various crops.
Answer 31:
Strong bases ionise completely in water, producing huge amounts of hydroxide ions (textrm{OH}^{-} ions). Generally, basic or alkali solutions of pH 8, 9 and 10 are considered weak bases or alkalis.
Weak bases ionise partially in water, producing less amounts of hydroxide ions (textrm{OH}^{-} ions).
Basic or alkali solutions of pH 11,12,13 and 14 are considered strong bases or alkalis.
Let us classify the given bases as strong or weak.
| Strong bases | Weak bases |
| Sodium hydroxide, NaOH Potassium hydroxide, KOH |
Ammonium hydroxide, NH4OH Calcium hydroxide, Ca(OH)2 Magnesium hydroxide, Mg(OH)2 |
Answer 32:
The ions present in the solutions of the following substances are given below.
(i) Hydrochloric acid, HCl = H+ and Cl– ions
(ii) Nitric acid, HNO3 = H+ and NO3– ions
(iii) Sulphuric acid, H2SO4 = 2H+ and SO4– ions
(iv) Sodium hydroxide, NaOH = Na+ and OH– ions
(v) Potassium hydroxide, KOH = K+ and OH– ions
(vi) Magnesium hydroxide, Mg(OH)2 = Mg2+ and 2OH– ions
Answer 33:
a. Pure water is a neutral substance with a pH of 7.
b. A universal indicator will show a green colour in a sugar solution (which is the colour for pH 7). An aqueous solution of sugar is also a neutral substance. Hence, its pH value is 7.
c. A universal indicator shows a greenish-yellow colour for pH 6 and orange yellow for pH 5. This means that if the indicator turns yellow, the pH should be between these two values. Hence, it must have a pH value between 5 and 6. As the pH is closer to 7 (between 5 and 6), it is a weak acid.
Answer 34:
(a) The pH in the stomach of a person suffering from indigestion would be less than 7, because of excess acid generation in the stomach.
(b) The pH of an antacid solution would be greater than 7. Antacids are anti-acids; that is, they are bases.
(c) Our stomach produces dilute hydrochloric acid, which helps in the digestion of food. Sometimes, excess acid is produced in the stomach, overeating being one of the causes. This results in indigestion along with pain. For relief, antacids, which are weak bases and neutralise the excess acid, are taken.
(d) Two common antacids are magnesium hydroxide (also called milk of magnesia) and sodium hydrogen carbonate (also called baking soda).
Answer 35:
On the pH scale, bases have values greater than 7, whereas acids have values less than 7. A basic solution turns red litmus blue. An acidic solution turns blue litmus red. With this information, let us separate the substances given below into those with pH values above and below 7.
| Substances | Acid/base | pH value | Litmus test result |
| 1.Lemon juice | Acid | Less than 7 | Turns blue litmus red |
| 2.Solution of washing soda | Base | Greater than 7 | Turns red litmus blue |
| 3. Toothpaste | Base | Greater than 7 | Turns red litmus blue |
| 4. Vinegar | Acid | Less than 7 | Turns blue litmus red |
| 5. Stomach juices | Acid | Less than 7 | Turn blue litmus red |
Answer 36:
(a) Yes, basic solutions also have H+ (aq) ions.
Generally, bases generate hydroxide ions when they are dissolved in water. Basic solutions also have H+ (aq) ions, which are obtained from the ionisation of water. The amount of H+ ions in basic solutions is very less compared with the amount of OH- ions. Hence, they are basic in nature.
(b) When a solution becomes more acidic, the pH becomes lower.
Answer 37:
(a) Acids: Substances that ionise on dissolving in water producing hydrogen ions (H+ions) are called acids. Acids are sour in taste and they turn blue litmus to red.
Examples: Hydrochloric acid, HCl, and sulphuric acid, H2SO4.
Bases: Substances that ionise on dissolving in water producing hydroxide ions (OH- ions) are called bases. Bases are bitter in taste, slippery to touch and they turn red litmus to blue.
Examples: Magnesium hydroxide, Mg(OH)2, and sodium hydroxide, NaOH.
(b) Two strong and two weak bases are the following.
| Strong bases | Formula | Weak bases | Formula |
| 1. Sodium hydroxide | NaOH | 1. Magnesium hydroxide | Mg(OH)2 |
| 2. Potassium hydroxide | KOH | 2. Calcium hydroxide | Ca(OH)2 |
(c) The types of ions formed are explained below.
(i) When an acid is dissolved in water, it ionises to produce hydrogen ions (H+ ions).
(ii) When a base is dissolved in water, it ionises to produce hydroxide ions (OH– ions).
(d) Let us consider an acid-base neutralisation reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH) to give sodium chloride salt (NaCl) and water.
Equations involving ions:Hydrochloric acid ionises in water as follows,
HCl(aq) → H+(aq) + Cl– (aq)
Sodium hydroxide ionises in water as
NaOH(aq) → Na+(aq) + OH–(aq)
Let us now combine both the reactions above.
Na+(aq) + Cl–(aq) + H+(aq) + OH–(aq) → NaCl(aq)+ H2O(l)
(e) The two important uses of bases are as follows.
1. Sodium hydroxide is widely used in the manufacture of soaps and detergents.
2. Magnesium hydroxide is used as an antacid, which gives relief from excess acidity in the stomach.
Exercise : Solution of Questions on page Number : 81
Answer 38:
(a) When zinc granules are heated with sodium hydroxide (base), sodium zincate salt is formed with the liberation of hydrogen gas.
Zn(s) + 2NaOH(aq) →Heat Na2ZnO2 (aq) + H2(g)
(b) Bases react with non-metal oxides to form a salt and water.
Bases + Non-metal oxides rightarrow Salt + Water
Example:
2NaOH(aq) + CO2(g) → Na2CO3(aq) + H2O(l)
Here, sodium hydroxide (base) reacts with carbon dioxide (non-metal oxide) to give sodium carbonate (salt) and water.
This reaction shows that non-metal oxides are acidic in nature.
Answer 39:
(a) Acids furnish hydrogen ions (H+ ions) when dissolved in water. The more will be the concentration of H+ ions in a solution, the lower will be its pH. Thus, an increase in the concentration of H+ ions in a solution leads to increase in the acidity of the solution.
(b) Bases furnish hydroxide ions (OH– ions) when dissolved in water. The concentration of hydroxide ions increases as bases dissolve in water. With the increase in the concentration on OH- ions, the pH also increases, thus, strengthening the basic nature of the solution.
(c) Universal indicator paper dipped into vinegar shows pH 3, indicating that vinegar is an acid. Acids show a value below 7 on the pH scale.
(d) Universal indicator paper put onto wet soap shows pH 8, indicating that wet soap is a base. Bases show a value above 7 on the pH scale.
(e) We know that solutions that have pH values less than 7 (from 0) are acidic, those with pH greater than 7 (up to 14) are basic (or alkaline) and those with pH 7 are neutral. The lower the pH, the higher the acidity, and the higher the pH, the higher the basicity. Let us classify solutions with the given pH values as acidic, alkaline or neutral.
| The pH of the solution | Nature of the solution |
| (i) 9 | Basic |
| (ii) 4 | Acidic |
| (iii) 7 | Neutral |
| (iv) 1 | Acidic |
| (v) 10 | Basic |
| (vi) 3 | Acidic |
Answer 40:
(c) Magnesium hydroxide
Magnesium hydroxide, which is a base, acts as an antacid. It neutralises excess acid present in the stomach and cures indigestion. Hence, it is used as a medicine.
Answer 41:
(a) an acidic solution
A bee sting contains an acidic solution. The name of the acid is methanoic acid.
Answer 42:
(d) an alkaline liquid
A wasp sting injects an alkaline solution into the skin, which causes high irritation and pain.
Answer 43:
(c) wasp
A honey bee, ant and a nettle leaf hair inject methanoic acid into the skin through their stings. But a wasp injects an alkaline solution into the skin through its sting.
Answer 44:
(d) 10
A basic solution turns red litmus blue. On the pH scale, bases have values greater than 7, whereas acids have values less than 7. Therefore, the pH of the given solution is likely to be 10.
Answer 45:
(b) 5
An acidic solution turns blue litmus red. On the pH scale, acids have values less than 7, whereas bases have values greater than 7. Solutions with pH equal to 7 are neutral in nature. Therefore, the pH of the given solution is likely to be 5.
Answer 46:
(c) 9
Every substance with a pH greater than 7 is considered basic or alkaline. Basic or alkaline solutions turn phenolphthalein indicator pink. Thus, the pH of the solution is most likely 9.
Answer 47:
(d) more than 7
Basic or alkaline solutions turn a methyl orange indicator yellow. Thus, the pH of the solution is likely to be more than 7.
Answer 48:
(b) sodium hydrogencarbonate
When a bee stings a person, it injects an acidic solution into the person’s skin, which causes severe pain and an itching sensation. Applying a baking soda solution (sodium hydrogencarbonate solution, which is a base) soothes the pain by neutralising the acid.
Answer 49:
(b) vinegar
When a wasp stings a person, it inserts an alkaline solution into the person’s skin. This causes high irritation and pain. Applying a weak acid, such as vinegar, to the wound neutralises the alkaline solution and soothes the pain.
Answer 50:
(a) wasp
When a wasp stings a person, it injects an alkaline solution into the person’s skin. This causes high irritation and pain. Applying a weak acid, such as vinegar, to the wound neutralises the alkaline solution and soothes the pain.
Answer 51:
(b) become less than 6
When fresh milk changes into curd, its acidity increases and it turns sour. Therefore, the pH of fresh milk becomes less than 6 when milk changes into curd.
Answer 52:
(c) hydrochloric acid
Our stomach produces dilute hydrochloric acid naturally. The acid that is produced, which has a pH of around 1.4, does not damage our stomach. Rather, it helps in the digestion of food.
Answer 53:
(b) lime
If the pH of the soil is 4.5, then it is highly acidic. Thus, soil should be treated with a base, such as quick lime (calcium oxide) or slaked lime (calcium hydroxide). These bases react with the excess acid present in the soil and neutralises it. Thus, the pH of the soil is increased, making it suitable for growing daffodils.
Exercise : Solution of Questions on page Number : 82
Answer 54:
(a) Usually, milk is acidic, with a pH of 6. The milkman has added a very small amount of baking soda to the milk to make it alkaline, so that it will not become sour with the formation of lactic acid. The alkaline medium prevents the milk from changing into curd. This is the reason why the milkman has shifted the pH of fresh milk from 6 to slightly alkaline.
(b) The milkman has made the milk alkaline by adding baking soda. The alkaline medium of milk slows down the transformation of the milk into curd. The lactic acid produced undergoes neutralisation, so the milk takes a long time to set as curd.
Answer 55:
Sulphur and Carbon burn in the air to form sulphur dioxide and carbon dioxide. Both are acidic oxides that indicate pH values less than 7 when moist pH paper is used.
S + O2 → SO2
C + O2 → CO2
Hydrogen is also a non-metal, but forms water, which has a neutral pH of 7, after reacting with oxygen. The oxides of magnesium and copper do not have pH values less than 7.
Answer 56:
(i) Solution D, with a pH value of 11, is weakly alkaline.
(ii) Solution C, with a pH value of 7, is neutral.
(iii) Solution A, with a pH value of 1, is strongly acidic.
(iv) Solution E, with a pH value of 13, is strongly alkaline.
(v) Solution B, with a pH value of 5, is weakly acidic.
Answer 57:
(a) Potatoes grow better in acidic soil.
(b) Broccoli grows better in alkaline soil.
Answer 58:
The solutions have been arranged below in the increasing order of their pH values.
Sulphuric acid (~ pH 1) < Car battery acid (~ pH 2) < Washing-up liquid (~ pH 5) < Milk of magnesia (~ pH 9) < Metal polish (~ pH 12) < Oven cleaner (~ pH 14)
Answer 59:
(a) As solution A is a basic solution, it would turn red litmus blue.
(b) As solution B is an acidic solution, it would turn blue litmus red.
(c) Sodium hydroxide and household ammonia can give solutions similar to solution A.
(d) Dilute hydrochloric acid and citric fruit juice can give solutions similar to solution B.
(e) When solution A (a base) reacts with solution B (an acid), a neutralisation reaction takes place, producing a salt and water.
Answer 60:
The information tells us that the chemical nature of
(a) a wasp sting is alkaline
(b) a bee sting is acidic
Answer 61:
(a) After each meal, sugar present in the food is broken down into acids by the bacteria present in the mouth. Acids have a pH less than 7, and as the acidity increases, the pH decreases. Thus, the pH in a person’s mouth becomes lower after each meal.
(b) When the pH in the mouth falls to around 5.5, tooth decay begins. As the acidity in the mouth increases, the acid reacts with the tooth enamel and causes tooth decay.
(c) A person can prevent tooth decay by consuming less sweet foods, such as ice creams and chocolates. Tooth decay can also be prevented by brushing with toothpastes (which is basic) regularly and by cleaning the mouth carefully after meals.
Answer 62:
(a) The students would have used universal indicator paper to measure the pH values of the substances. Universal indicator paper indicates different pH values of solutions with different colour changes. The strength of an acid or a base is known with the help of different colours.
(b) Lemon juice, with pH 2.5, is most acidic.
(c) Household ammonia, with pH 12.0, is most alkaline.
(d) Salt and sugar, with pH 7, are neutral.
(e) Vinegar, with pH 3, can be used to treat wasp stings. As a wasp sting is alkaline (pH > 7), an acid, such as vinegar, is applied to the skin, and it neutralises the base and soothes the pain.
(f) Baking soda, with pH 8.5, can be used to treat bee stings. As a bee sting is acidic (pH < 7), a base, such as baking soda, is applied to the skin, and it neutralises the acid and soothes the pain.
Exercise : Solution of Questions on page Number : 83
Answer 63:
(a) X is the metal zinc (Zn) and Y is hydrogen gas.
Zinc reacts with hydrochloric acid and forms zinc chloride salt with the liberation of hydrogen gas (Y), which burns with a ‘pop’ sound. Zinc displaces hydrogen from hydrochloric acid.
Hence, X is zinc and Y is hydrogen.
(b) The chemical equation is as follows.
(i) Zn + 2HCl →ZnCl2 + H2 ↑
(ii) 2NaOH + Zn →Na2ZnO2 + H2
Exercise : Solution of Questions on page Number : 96
Answer 1:
(a) The chemical formula of baking soda is NaHCO3. Its chemical name is sodium hydrogen carbonate.
(b) The chemical formula of washing soda is Na2CO3.10H2O. Its chemical name is sodium carbonate decahydrate.
Answer 2:
(i) The chemical formula of soda ash is Na2CO3. Its chemical name is sodium carbonate.
(ii) The chemical formula of sodium carbonate decahydrate is Na2CO3.H2O. Its common name is washing soda.
Answer 3:
False
Water of crystallisation is an integral part of the crystal structure of salts. It is not free or additional water outside the crystal; thus, salts do not appear wet. They seem to be completely dry.
Answer 4:
CuSO4.H2O has a blue colour.
The blue colour is due to the presence of water molecules in the structure. The colour of a salt crystal is usually given by the water of crystallisation. But anhydrous salts (without the water molecules) lose their colour and shape and become white. Thus, CuSO4, which does not have water molecules, appears white.
Answer 5:
Litmus turns blue in a solution of sodium carbonate.
Answer 6:
When plaster of Paris is mixed with water, it settles into a hard mass to form gypsum (common name). The chemical name of gypsum is calcium sulphate dihydrate.
CaSO4.12H2O + 112H2O → CaSO4.2H20.
Answer 7:
Chlorine should be treated with dry slaked lime (calcium hydroxide) to get bleaching powder (calcium oxychloride).
The reaction can be given as follows.
Ca(OH)2 + Cl2 →CaOCl2 + H2O
Answer 8:
The commercial name of calcium sulphate hemihydrate is plaster of Paris. The formula is CaSO4.12H2O.
Exercise : Solution of Questions on page Number : 97
Answer 9:
When hydrogen and chlorine react with each other, hydrochloric acid (textrm{HCl}) is formed.
H2 + Cl2 → 2HCl
Answer 10:
Calcium sulphate hemihydrate, popularly known as plaster of Paris, is a calcium compound that hardens on wetting with water. Its formula is CaSO4.12H2O.
Answer 11:
Sodium carbonate decahydrate (Na2CO3.10H2O), popularly known as washing soda, is a constituent of many dry soap powders.
Answer 12:
Washing soda (sodium carbonate decahydrate) is soluble in water. It has the formula Na2CO3 .10H2O.
Answer 13:
Tartaric acid is present in baking powder, which is a mixture of tartaric acid and sodium hydrogen carbonate.
Answer 14:
Sodium is the metal whose carbonate, sodium carbonate decahydrate, is known as washing soda.
Answer 15:
NaHCO3.
Sodium hydrogen carbonate (baking soda) is used as an antacid, to remove acidity in the stomach. As baking soda is alkaline, it reacts with excess acid and neutralises the acid.
Answer 16:
a. NaHCO3 is commonly known as baking soda.
b. Na2CO3.10H2O is commonly known as washing soda.
Answer 17:
a. The chemical name of common salt is sodium chloride. Its formula is NaCl.
b. The chemical name of caustic soda is sodium hydroxide. Its formula is NaOH.
Answer 18:
Common salt occurs naturally in sea water in the dissolved form.
Common salt is found in underground deposits as rock salt.
Answer 19:
Common salt (or sodium chloride) is present naturally in sea water in the dissolved form. This is the major salt present in sea water.
Answer 20:
Common salt is obtained from sea water by the process of evaporation through the sun’s heat, which leaves behind crude common salt. Crude salt is further purified to remove impurities as it has some other salts mixed with it.
Answer 21:
Sodium chloride is required in our body for the functioning of our nervous system, the movement of muscles, the production of hydrochloric acid in the stomach for digestion of food and various other biochemical processes which occur in our body.
Answer 22:
Chemicals made from common salt:
Sodium hydroxide (NaOH), or caustic soda
Washing soda (Na2CO3.10H2O), or sodium carbonate decahydrate
Baking soda (NaHCO3), or sodium hydrogen carbonate.
Answer 23:
Two uses of common salt are as follows.
It is used in the manufacture of soap.
It is used to melt ice that collects on the roads in cold countries during winter.
Answer 24:
The common salt mined from underground deposits is called rock salt.
When ancient seas dried up by evaporation thousands of years ago, rock salt, which we dig out today, was formed.
Answer 25:
Common salt (NaCl) is used as a preservative in pickles and in the curing (or preservation) of meat and fish.
Answer 26:
Common salt (or sodium chloride) is the raw material for making caustic soda. The chemical formula of sodium chloride is NaCl.
Answer 27:
Three products obtained by the electrolysis of an aqueous solution of sodium chloride are chlorine gas (formed at the anode), hydrogen gas (formed at the cathode) and sodium hydroxide solution (formed near the cathode).
Answer 28:
During the electrolysis of a saturated solution of sodium chloride,
(a) chlorine gas is produced at the anode (positive electrode);
(b) hydrogen gas is produced at the cathode (negative electrode);
(c) sodium hydroxide solution is produced near the cathode.
Answer 29:
(a) Common salt is obtained from sea-water by the process of evaporation.
(b) Rock salt is mined just like coal.
(c) Chemical formula of washing soda is Na2CO3.10H2O.
(d) Sodium hydrogen carbonate is baking soda whereas sodium carbonate is washing soda.
(e) The chemical formula of plaster of Paris is CaSO4.1/2H2O.
Answer 30:
(a) 2NaCl(aq) + 2H2O(l) →Electricity 2NaOH(aq) +Cl2(g) +H2(g)(b) 2NaHCO3 →Heat Na2CO3 + CO2 + H2O(c) NaCl + NH3 + H2O + CO2 →NaHCO3 +NH4Cl(d) Ca(OH)2 + Cl2 → CaOCl2 +H2O
Answer 31:
Washing soda is sodium carbonate. It contains 10 molecules of water of crystallisation and is also known as sodium carbonate decahydrate. The chemical formula is Na2CO3.10H2O.
Properties:
Washing soda is a transparent crystalline solid.
Washing soda is soluble in water. The solution of washing soda is alkaline and turns red litmus paper to blue.
Uses:
Washing soda is used for removing permanent hardness of water.
It is used in the manufacture of soap, glass, paper and sodium compounds, such as borax.
Answer 32:
The formula of sodium chloride is NaCl and that of sodium carbonate is Na2CO3.
An aqueous solution of sodium chloride is neutral because sodium chloride is formed from a strong acid, hydrochloric acid (HCl), and a strong base, sodium hydroxide(NaOH). When sodium chloride is dissolved in water, it gets hydrolysed to give equal amounts of hydroxide and hydrogen ions, and this makes its aqueous solution neutral.
NaCl(s) + H2O(l) → NaOH(aq) + HCl(aq)
An aqueous solution of sodium carbonate (washing soda) is alkaline because it is a salt prepared from the reaction of a weak acid, carbonic acid (H2CO3), and a strong base, sodium hydroxide (NaOH). When sodium carbonate is dissolved in water, it gets hydrolysed to some extent and forms sodium hydroxide and carbonic acid.
Na2CO3(s) + 2H2O(l) → 2NaOH(aq) + H2CO3(aq)
Sodium hydroxide, being a strong base, is fully ionised to give a large amount of hydroxide ions (OH–), and carbonic acid, being a weak acid, is slightly ionised to form a small amount of hydrogen (H+) ions. Therefore, an aqueous solution contains more hydroxide ions than hydrogen ions, and this makes the solution basic.
Answer 33:
The chemical formula of ammonium chloride is NH4Cl. It is formed from a weak base, ammonium hydroxide (NH4OH), and a strong acid, hydrochloric acid (HCl). On dissolving in water, NH4Cl hydrolyses to some extent to form ammonium hydroxide (NH4OH) and hydrochloric acid (HCl).
NH4Cl(s) + H2O(l) → NH4OH(aq)Weak Base+HCl(aq)Strong Acid
Hydrochloric acid is a strong acid that is fully ionised to give a large amount of H+ (hydrogen) ions and ammonium hydroxide is a weak base that is slightly ionised to give a small amount of OH– (hydroxide) ions. As ammonium chloride contains more H+ ions than OH- ions, it is acidic with a pH less than 7.
Answer 34:
Baking soda is a sodium salt used for speeding up the cooking of foodstuff such as various pulses. The chemical name of baking soda is sodium hydrogencarbonate (NaHCO3), also called sodium bicarbonate.
Uses:
Baking soda is used in fire extinguishers.
It is an ingredient, used in preparing baking powder, which is used in baking cakes, breads, etc.
It is used as an antacid to remove acidity in the stomach.
Washing soda, or sodium carbonate (Na2CO3.10H2O), consists of two sodium atoms attached to a carbonate group with 10 molecules of water of crystallisation, whereas baking soda comprises one atom of sodium and one of hydrogen, attached to a carbonate group. Hence, baking soda (NaHCO3) differs from washing soda (Na2CO3) by one hydrogen atom. When baking soda is heated up at high temperatures, it breaks down to form sodium carbonate, water vapour and carbon dioxide.
Answer 35:
Sodium hydrogencarbonate (baking soda) is produced on a large scale by reacting a cold and concentrated solution of sodium chloride called brine with ammonia and carbon dioxide.
NaCl + NH3 + H2O + CO2 → NaHCO3 + NH4Cl
Answer 36:
Sodium hydrogencarbonate (baking soda) is produced when a cold and concentrated solution of sodium chloride (brine) reacts with ammonia and carbon dioxide.
NaCl + NH3 + H2O + CO2 → NaHCO3 + NH4Cl
Exercise : Solution of Questions on page Number : 98
Answer 37:
(a) The water molecules that form part of the structure of a crystal of a salt are called water of crystallisation. For example, washing soda crystals (or sodium carbonate crystals) contain 10 molecules of water of crystallisation in one formula unit. It is represented as, Na2CO3.10H2O. It is also called sodium carbonate decahydrate the last part of name coming from ten molecules of water present in the crystal structure.
(b) When copper sulphate crystals (chemical formula CuSO4.5H2O) are heated strongly, they lose all the water of crystallisation and form anhydrous copper sulphate (CuSO4), which is white. As the blue colour of the crystals disappeared on heating, we can conclude that the colour was due to water of crystallisation. This proves that blue copper sulphate crystals contain water of crystallisation.
CuSO4.5H2O → CuSO4 + 5H2O
(c) A few drops of the liquid to be tested are added to white anhydrous copper sulphate powder. The appearance of a blue colour in anhydrous copper sulphate indicates the presence of moisture or water in the liquid.
Answer 38:
(a) The common name of sodium hydrogencarbonate is baking soda.
(b) When a solution of sodium hydrogen carbonate (NaHCO3) is heated, it decomposes to give sodium carbonate (Na2CO3) with the evolution of carbon dioxide gas.
2NaHCO3→Heat Na2CO3+ CO2+ H2O
(c) Sodium hydrogen carbonate is used as an antacid because it is alkaline and it neutralises excess acid present in the stomach and relieves indigestion.
Answer 39:
(a) If gypsum (or calcium sulphate dihydrate) is heated above 100⁰C (373K) while preparing plaster of Paris, then all its water of crystallisation is eliminated and anhydrous calcium sulphate (CaSO4), called dead burnt plaster, is formed. The anhydrous calcium sulphate does not set like plaster of Paris on addition of water.
(b) CaSO4·12 H2O + 32H2O →CaSO4·2H2O.
Answer 40:
(a) When copper sulphate (CuSO4.5H2O) crystals are heated strongly, they lose all the water of crystallisation and form anhydrous copper sulphate, which is white.
CuSO4.5H2O → CuSO4 + 5H2O
(b) Anhydrous copper sulphate gets hydrated and turns blue on addition of a few drops of water.
CuSO4 + 5H2O → CuSO4.5H2O .
Answer 41:
(a) Two constituents of baking powder are baking soda (sodium hydrogencarbonate) and tartaric acid.
(b) Baking powder is a mixture of baking soda (sodium hydrogencarbonate, NaHCO3) and a solid edible acid such as tartaric acid, whereas baking soda is a single compound (sodium hydrogencarbonate, NaHCO3).
(c) On mixing baking powder with water, which is present in the dough prepared for baking a cake or bread, sodium hydrogencarbonate reacts with tartaric acid to evolve carbon dioxide gas. The carbon dioxide gas gets trapped in the wet dough and bubbles out slowly, and the cake becomes soft and spongy.
NaHCO3(aq) + H+(aq) → Na+(aq) + CO2(g) + H2O(l)
Note: Here, H+ is from tartaric acid and Na+ is from sodium tartrate salt.
Answer 42:
(a) The chemical name of bleaching powder is calcium oxychloride.
(b) The chemical formula of bleaching powder is CaOCl2.
(c) The substances used for the preparation of bleaching powder are chlorine (Cl2) and slaked lime [Ca(OH)2].
(d) Bleaching powder is used as a disinfectant. It is used for making drinking water free from germs.
Answer 43:
A soda-acid type of fire extinguisher contains solutions of sulphuric acid and sodium hydrogencarbonate in separate containers.
The working of a soda-acid fire extinguisher is described below:
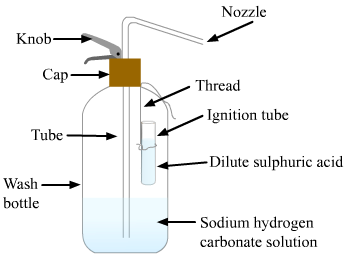
Answer 44:
(a) Sodium carbonate or washing soda (Na2CO3) is used for softening hard water.
(b) Bleaching powder or calcium oxychloride (CaOCl2) is used for disinfecting drinking water supply.
(c) Sodium carbonate, or washing soda (Na2CO3), has a cleansing property.
(d) Bleaching powder, or calcium oxychloride (CaOCl2), is used for removing the colour of a coloured cloth.
(e) Plaster of Paris has a remarkable property of setting into a hard mass on addition of water.
(f) Bleaching powder, or calcium oxychloride (CaOCl2), is obtained by the action of chlorine on solid (dry) slaked lime.
Answer 45:
(a) Gypsum is calcium sulphate dihydrate (CaSO4.2H2O). When gypsum is heated to a temperature of 100⁰C (373K), it loses three-fourths of its water of crystallisation and forms plaster of Paris (CaSO4.1/2H2O).
(b) Sodium carbonate or washing soda is used for making borax and glass.
(c) Plaster of Paris sets into a hard mass on addition of water. It is easy to mould a layer Plaster of Paris around the damaged part, which later solidifies and provides support to the damaged bone for its quick healing. This is why it is used in hospitals for setting fractured bones.
(d) Chlorine is the real bleaching agent present in bleaching powder.
Answer 46:
(a) Baking powder is a sodium salt called sodium bicarbonate (NaHCO3). It is a mixture of baking soda (sodium hydrogencarbonate) and a mild edible acid such as tartaric acid. When baking soda mixes with water, the sodium hydrogencarbonate reacts with the tartaric acid to produce carbon dioxide gas. The carbon dioxide gas produced gets trapped in the wet dough made for baking a cake or bread, and bubbles out slowly, and the cake or bread becomes soft and spongy.
(b) In addition to sodium hydrogencarbonate, baking powder contains tartaric acid (X). Tartaric acid is added to baking powder to neutralise the bitter taste that baking soda produces in the cake. Also, carbon dioxide, which makes the cake fluffy and soft, is produced only when the tartaric acid reacts with the sodium hydrogencarbonate or baking soda, in the presence of water.
Answer 47:
(a) Sodium hydroxide:
(i) It is used for making soaps and detergents.
(ii) It is used for making synthetic textile fibre (such as rayon).
(b) Chlorine:
(i) It is used for the production of bleaching powder.
(ii) It is used to sterilise drinking water supply and the water in swimming pools, because it is a disinfectant.
(c) Hydrogen:
(i) It is used in the hydrogenation of oils to obtain solid fats called vegetable ghee.
(ii) Liquid hydrogen is used as a fuel for rockets.
(d) Hydrochloric acid:
(i) It is used for cleaning iron sheets before tin plating or galvanisation.
(ii) It is used in medicines and cosmetics.
Answer 48:
(a)The common name of the compound CaOCl2 is bleaching powder.
(b) The raw material used for the preparation of plaster of Paris is gypsum (CaSO4.2H2O).
(c) Plaster of Paris has a property of setting into a hard mass in about half an hour after wetting with water. This property is utilised in making casts for broken limbs in hospitals.
(d) Chlorine is a disinfectant used for sterilising drinking water supply in order to kill disease-causing bacteria that the water or water pipes might contain.
Answer 49:
(a) When electricity is passed through a concentrated solution of sodium chloride called brine, the solution decomposes to form sodium hydroxide, chlorine and hydrogen.
2 NaCl(aq) + 2 H2O(l) → electrolysiselectrolyte 2 NaOH(aq) + Cl2(g) + H2(g)
(b) The process of electrolysis of sodium chloride solution is called the chlor-alkali process because ‘chlor’ (chlorine) and alkali (sodium hydroxide, NaOH) are formed as products.
(c) The three main products of the chlor-alkali process are H2, Cl2 and NaOH.
Uses of sodium hydroxide:
(i) It is used for making soaps and detergents.
(ii) It is used for making synthetic textile fibre (such as rayon).
Uses of chlorine:
(i) It is used in the production of bleaching powder.
(ii) It is used to sterilise drinking water supply and the water in swimming pools, because it is a disinfectant.
Uses of hydrogen:
(i) It is used in the hydrogenation of oils to obtain solid fats called vegetable ghee.
(ii) Liquid hydrogen is used as a fuel for rockets.
Answer 50:
Preparation of washing soda:
A cold and concentrated solution of sodium chloride is reacted with ammonia (NH3) and carbon dioxide to form sodium hydrogen carbonate (NaHCO3) and ammonium chloride (NH4Cl). Sodium hydrogen carbonate precipitates as a solid as it is slightly soluble in water.
NaCl + NH3 + H2O + CO2 → NaHCO3 + NH4Cl
The sodium hydrogen carbonate precipitated is separated by filtration, and then dried and heated. On heating, it decomposes to form sodium carbonate (Na2CO3). The anhydrous sodium carbonate formed here is called soda ash.
2NaHCO3 → Na2CO3 + CO2+ H2O
The soda ash is dissolved in water and recrystallised to get washing soda crystals containing 10 molecules of water of crystallisation.
Na2CO3 + 10H2O → Na2CO3.10H2O
An aqueous solution of washing soda is alkaline, because sodium carbonate is a salt prepared from the reaction between a weak acid, carbonic acid (H2CO3), and a strong base, sodium hydroxide (NaOH). When sodium carbonate is dissolved in water, it gets hydrolysed to some extent and forms sodium hydroxide and carbonic acid.
Na2CO3(s) + 2H2O(l) → 2NaOH(aq) + H2CO3(aq)
Sodium hydroxide, being a strong base, is fully ionised to give a large amount of hydroxide ions (OH–), and carbonic acid, being a weak acid, is slightly ionised to form a small amount of hydrogen (H+) ions. Therefore, an aqueous solution contains more hydroxide ions than hydrogen ions, which makes the solution basic.
By saying that washing soda has detergent properties, or cleansing properties, one means that it can remove dirt and grease from dirty clothes, etc. It attacks dirt and grease to form water-soluble products, which are washed away on rinsing with water.
Uses of washing soda:
It is used for removing permanent hardness of water.
It is used in the manufacture of glass, soap, paper and sodium compounds, such as borax.
Answer 51:
Bleaching powder is calcium oxychloride (CaOCl2) and is also called chloride of lime.
Preparation:
Bleaching powder is prepared by passing chlorine gas over dry slaked lime.
Ca(OH)2 + Cl2 → CaOCl2 + H2O
(Slaked lime)
When bleaching powder reacts with dilute sulphuric acid, all the chlorine present in it is liberated. This chlorine acts as a bleaching agent.
CaOCl2 + H2SO4 → CaSO4 + Cl2 + H2O
c. Uses:
i. Bleaching powder is used in making chloroform (CHCl3).
ii. It is used as an oxidising agent in many chemical industries.
Answer 52:
(a) Plaster of Paris is calcium sulphate hemi-hydrate (calcium sulphate half hydrate). The chemical formula of plaster of Paris is CaSO4.1/2H2O.
(b) When gypsum (CaSO4.2H2O) is heated to a temperature of 100⁰C (373K), it loses three-fourths of its water of crystallisation and forms plaster of Paris.
CaSO4.2H2O →∆ CaSO4.1/2H2O + 3/2 H2O
Gypsum Plaster of Paris
(c) Plaster of Paris should be stored in a moisture-proof container because the presence of moisture can cause its slow setting by the addition of water of hydration, and it would be useless after some time.
(d) Uses:
(i) Plaster of Paris is used in making decorative materials, toys, cosmetics, cheap ornaments, black-board chalks and casts for fractured bone and statues.
(ii) It is used in making surfaces such as walls of houses smooth before they are painted and for creating ornamental designs on the ceilings of houses and other buildings.
Answer 53:
Salt is a compound formed when a metal replaces the hydrogen atom in an acid. The names and formulae of two salts are sodium chloride, NaCl, and ammonium chloride, NH4Cl, respectively. Sodium chloride salt (NaCl) is formed from an acid, hydrochloric acid (HCl), and a base (NaOH). Ammonium chloride (NH4Cl) is formed from an acid, hydrochloric acid (HCl), and a base, ammonium hydroxide (NH4OH).
Salts having the same positive ions or the same negative ions belong to one family of salts. For example, calcium chloride (CaCl2) and calcium sulphate (CaSO4) belong to the same family of salts called ‘calcium salts’ because they both contain the same positively charged ions, calcium ions (Ca2+). Similarly, zinc sulphate (ZnSO4) and copper sulphate (CuSO4) belong to the same family of salts called ‘sulphate salts’ because they both contain the same negatively charged ions, sulphate ions (SO42-).
Salts that contain water of crystallisation are called ‘hydrated salts’. For example, copper sulphate crystals contain five molecules of water of crystallisation in one formula unit and hence is written as CuSO4.5H2O. It is also called copper sulphate pentahydrate.
Salts that have lost their water of crystallisation on heating are called ‘anhydrous salts’. For example, when copper sulphate crystals (CuSO4.5H2O), which are blue, are heated strongly, they lose all the water of crystallisation and form anhydrous copper sulphate (CuSO4), which is white.
Hydrated salts:
Copper sulphate pentahydrate crystals, CuSO4.5H2O, are blue.
Iron sulphate heptahydrate crystals, FeSO4.7H2O, are green.
Blue litmus turns red. NH4Cl is formed from a weak base, ammonium hydroxide (NH4OH), and a strong acid, hydrochloric acid (HCl). So, on dissolving in water, NH4Cl will hydrolyse to form more H+ ions than OH– ions, and thus it is acidic.
Answer 54:
b) NH4Cl
NH4Cl is formed with a weak base, ammonium hydroxide (NH4OH), and a strong acid, hydrochloric acid (HCl). On dissolving in water, NH4Cl hydrolyses to form more H+ ions than OH– ions. Thus, it is acidic with a pH less than 7.
Answer 55:
(a) Na2CO3
Na2CO3 is formed from a weak acid, carbonic acid (H2CO3), and a strong base, sodium hydroxide (NaOH). On dissolving in water, Na2CO3 hydrolyses to form more OH– ions than H+ ions. Therefore, its solution in water is basic, or alkaline, with a pH more than 7.
Answer 56:
(c) KCl
KCl is formed from a strong acid, hydrochloric acid (HCl), and a strong base, potassium hydroxide (KOH). On dissolving in water, KCl hydrolyses to form equal amounts of H+ and OH– ions. This is why an aqueous solution of KCl is neutral with a pH of almost 7.
Answer 57:
b) H2, Cl2 and NaOH
The process of electrolysis of sodium chloride solution is called the chlor-alkali process because of the products formed, i.e., H2, Cl2 and NaOH. ‘Chlor’ stands for chlorine and ‘alkali’ for sodium hydroxide (NaOH).
Answer 58:
c) ten
The chemical formula of washing soda is Na2CO3.10H2O. The water molecules that form part of the structure of a salt are called its water of crystallisation. Hence, the number of molecules of water of crystallisation present in washing soda crystals is 10.
Answer 59:
a). ammonium sulphate
Ammonium sulphate [(NH4)2SO4] is formed from a weak base, ammonium hydroxide (NH4OH), and a strong acid, sulphuric acid (H2SO4). On dissolving in water, (NH4)2SO4 hydrolyses to form more H+ ions than OH– ions. As the aqueous solution of the salt (NH4)2SO4 is acidic, it turns blue litmus red.
Answer 60:
(d) potassium carbonate
Potassium carbonate (K2CO3) is formed from a weak acid, carbonic acid (H2CO3), and a strong base, potassium hydroxide (KOH). On hydrolysis, K2CO3 forms more OH– ions than H+ ions. Thus, it is basic and therefore turns red litmus blue.
Answer 61:
a) potassium sulphate
Potassium sulphate (K2SO4) is formed from a strong acid, sulphuric acid (H2SO4), and a strong base, potassium hydroxide (KOH). On dissolving in water, K2SO4 hydrolyses to form equal amounts of H+ and OH- ions. As the salt solution of K2SO4 is neutral, it has no effect on red or blue litmus paper.
Answer 62:
(c) K2CO3
K2CO3 is formed from a weak acid, carbonic acid (H2CO3), and a strong base, potassium hydroxide (KOH). On hydrolysis, K2CO3 forms more OH– ions than H+ ions, and thus, it is basic. As the aqueous solution of the salt is basic, it turns a phenolphthalein indicator pink.
Answer 63:
(c) NaHCO3
The chemical name of baking soda is sodium hydrogen carbonate, and its chemical formula is NaHCO3.
Answer 64:
(b) Ca(OH)2
Bleaching powder (CaOCl2) is formed by passing chlorine gas over slaked lime [Ca(OH)2].
Answer 65:
(d) CaSO4.2H2O
When CaSO4.2H2O (gypsum) is heated to a temperature of 100⁰C (373K), it loses three-fourths of its water of crystallisation and forms plaster of Paris (CaSO4.1/2H2O).
Answer 66:
(a) K2CO3
K2CO3 is formed from a weak acid, carbonic acid (H2CO3), and a strong base, potassium hydroxide (KOH). On hydrolysis, K2CO3 forms more OH– ions than H+ ions. Thus, it is basic, with a pH more than 7.
Answer 67:
(d) NH4Cl
NH4Cl is formed from a weak base, ammonium hydroxide (NH4OH), and a strong acid, hydrochloric acid (HCl). On hydrolysis, NH4Cl forms more H+ ions than OH– ions. Thus, it is acidic, with a pH less than 7.
Answer 68:
(d) KCl
KCl is formed from a strong acid, hydrochloric acid (HCl), and a strong base, potassium hydroxide (KOH). Thus, KCl, on hydrolysis, forms equal amounts of H+ and OH– ions. This is why an aqueous solution of KCl shows a pH of almost 7.
Exercise : Solution of Questions on page Number : 100
Answer 69:
a) Neither solution will turn blue litmus red.
Solution P is sodium chloride, formed from a strong acid, hydrochloric acid (HCl), and a strong base, sodium hydroxide (NaOH). When sodium chloride is dissolved in water, it gets hydrolysed to give equal amounts of hydroxide and hydrogen ions, which makes its aqueous solution neutral. Therefore, the solution of sodium chloride will have no effect on red or blue litmus paper.
b) Sodium hydroxide solution (Q) will turn red litmus blue, because solution Q is basic.
Answer 70:
Copper sulphate crystals (A) are blue and have the chemical formula CuSO4.5H2O. When copper sulphate crystals are heated strongly, they lose all the water of crystallisation (B) and form anhydrous copper sulphate (C), which is white.
A C B
CuSO4.5H2O → CuSO4 + 5H2O
Anhydrous copper sulphate turns blue on addition of a few drops of water (D) because it gets hydrated again.
C D
CuSO4 + 5H2O → CuSO4.5H2O
Answer 71:
Substance X, which gives sodium hydroxide, Cl2 and H2 upon electrolysis, is NaCl. The electrolysis of sodium chloride solution is called the chlor-alkali process because of the products formed, ‘chlor’ for chlorine and ‘alkali’ for sodium hydroxide (NaOH).
Answer 72:
a) Ca(OH)2 combines with Cl2 to give bleaching powder (CaOCl2 ).
The chemical reaction involved is as follows.
2Ca(OH)2 + CaCl2 → CaOCl2 + 2H2O
b) NaCl, NH3, H2O, CO2 are utilised in the production of washing soda.
The chemical reaction involved can be shown in three steps:
i) Production of sodium hydrogen carbonate:
NaCl + NH3 + H2O + CO2 → NaHCO3 +NH4Cl
ii) Production of anhydrous sodium carbonate upon heating sodium hydrogen carbonate:
2NaHCO3 → Na2CO3 + CO2 + H2O
iii) Production of hydrated sodium carbonate when anhydrous sodium carbonate is dissolved in water:
Na2CO3 + 10H2O → Na2CO3.10H2O
c) 2CaSO4.H2O represents plaster of Paris as its chemical formula is CaSO4.1/2H2O.
d) NaHCO3 is part of baking powder, as baking powder is a mixture of NaHCO3 and tartaric acid.
e) NaHCO3 is used as an antacid, as it is alkaline, thus it neutralises excess acid in the stomach.
Answer 73:
a) NH4Cl is formed from a weak base, ammonium hydroxide (NH4OH), and a strong acid, hydrochloric acid (HCl). On hydrolysis, NH4Cl forms more H+ ions than OH– ions. Thus, it is acidic, with a pH less than 7.
b) KCl is formed from a strong acid, hydrochloric acid (HCl), and a strong base, potassium hydroxide (KOH). Thus, KCl, on hydrolysis, forms equal amounts of H+ and OH– ions. Therefore, an aqueous solution of KCl is neutral, with a pH of almost 7.
c) K2CO3 is formed from a weak acid, carbonic acid (H2CO3), and a strong base, potassium hydroxide (KOH). On hydrolysis, K2CO3 forms more OH– ions than H+ ions. Thus, it is basic, with a pH more than 7.
Answer 74:
a) Compound X is plaster of Paris, or calcium sulphate half hydrate (CaSO4.1/2H2O)
b) CaSO4.2H2O → CaSO4.1/2H2O + 3/2 H2O
c) Plaster of Paris is used in hospitals for setting fractured bones in the right position to ensure correct healing. Casts made of plaster of Paris help keep the fractured bone straight.
Answer 75:
(a) The salt solutions of NH4Cl and (NH4)2SO4 give acidic solutions, because these salts are each formed from a weak base and a strong acid. The ionisation of a weak base is not complete, whereas a strong acid ionises completely. Hence, there are more H+ ions than OH- ions, making the solutions acidic.
(b) The salt solutions of NaCl and K2SO4 are neutral, because these salts are formed from a strong base and a strong acid, and on ionisation, there is an equal number of H+ and OH– ions, making the solutions neutral.
(c) The salt solutions of CH3COONa and Na2CO3 are basic, because these salts are each formed from a weak acid and a strong base. The ionisation of a weak acid is not complete, whereas a strong base ionises completely. Hence, there are more OH– ions than H+ ions, making the solutions basic.
Answer 76:
Bleaching powder is a white powder that emits a strong smell of chlorine and is used for disinfecting drinking water supply at waterworks.
The chemical name of bleaching powder is calcium oxychloride (CaOCl2).
The chemical reaction that takes place in its preparation is as follows.
Ca(OH)2 + Cl2 → CaOCl2 + H2O
Answer 77:
As the aqueous solution of salt X turns red litmus blue, it must be basic in nature.
For example, an aqueous solution of sodium acetate salt is basic. Sodium acetate (CH3COONa) is a salt prepared from the reaction between a weak acid, acetic acid (CH3COOH), and a strong base, sodium hydroxide (NaOH). When sodium acetate is dissolved in water, it gets hydrolysed to some extent to form sodium hydroxide and acetic acid.
CH3COONa (s) + H2O(l) → NaOH(aq) + CH3COOH(aq)
Sodium hydroxide, being a strong base, is fully ionised to give a large amount of hydroxide ions (OH–), and acetic acid, being a weak acid, is slightly ionised to form a small amount of hydrogen (H+) ions. Therefore, the aqueous solution contains more hydroxide ions than hydrogen ions, which makes the solution basic.
Answer 78:
The person forgot to add baking powder.
Baking powder is a mixture of baking soda (sodium hydrogencarbonate) and a mild edible acid such as tartaric acid.
When baking powder mixes with water, the sodium hydrogen carbonate reacts with the tartaric acid to emit carbon dioxide gas. The carbon dioxide gas produced gets trapped in the wet dough made for baking a cake or bread, and bubbles out slowly. Thus, the cake becomes soft and spongy.
Answer 79:
Plaster of Paris, or calcium sulphate hemi hydrate (CaSO4.1/2H2O), is a white compound that sets into a hard mass in about half an hour after wetting with water. This property of plaster of Paris is used in making casts for broken limbs in hospitals.
Answer 80:
When chlorine and sodium hydroxide produced during the electrolysis of brine are allowed to mix, a new chemical called sodium hypochlorite (NaClO) is formed.
Sodium hypochlorite is a bleaching agent that is used in making household bleaches and for bleaching fabrics.
Answer 81:
Gypsum (calcium sulphate crystals) contains two molecules of crystallisation in one formula unit and hence is written as CaSO4.2H2O. It is also called calcium sulphate dihydrate.
Copper sulphate crystals contain five molecules of crystallisation in one formula unit. The chemical formula is CuSO4.5H2O (copper sulphate pentahydrate).
Washing soda crystals (sodium carbonate crystals) contain 10 molecules of crystallisation in one formula unit. The chemical formula is Na2CO3.10H2O (sodium carbonate decahydrate).
Answer 82:
a) Copper sulphate crystals contain five molecules of crystallisation in one formula unit. The chemical formula is CuSO4.5H2O (copper sulphate pentahydrate) .
b) Washing soda crystals (sodium carbonate crystals) contain 10 molecules of crystallisation in one formula unit. The chemical formula is Na2CO3.10H2O (sodium carbonate decahydrate).
c) Gypsum (calcium sulphate crystals) contains two molecules of water of crystallisation in one formula unit. The chemical formula is CaSO4.2H2O (calcium sulphate dihydrate).
